Testimonials
We experienced strong purity and activity differences between in-house produced Tn5 batches and switched to buying Tn5 from Diagenode with higher activity and small batch effects only.
Rebekka Scholz et al. Combined Analysis of mRNA Expression and Open Chromatin in Microglia. Methods Mol Biol. 2024;2713:543-571.
about Tagmentase (Tn5 transposase) unloaded, Tagmentase (Tn5 transposase) unloaded, Tagmentase (Tn5 transposase) unloaded, Tagmentase (Tn5 transposase) unloaded, Tagmentase (Tn5 transposase) unloaded.
We are really happy to use the loaded pA-Tn5 for CUT&Tag. Easy to use and reproductible for samples with a starting low number of cells. Besides, Diagenode has a great customer service and technical support which are really responsive.
Cathy NGUYEN, CNRGH, Evry-Courcouronnes, France.
about pA-Tn5 Transposase - loaded, pA-Tn5 Transposase - loaded, pA-Tn5 Transposase - loaded.
We have been using the Hyperactive Tagmentase for 2 years and its performance is outstanding - short operation time and good reproducibility, outmatching the competition. Moreover the interaction with customer representatives is always top-notch - highly efficient and knowledgeable. I can't recommend enough!
Julia Liz Touza, AstraZeneca Gothenburg, Sweden
about Tagmentase (Tn5 transposase) - loaded - replaced by the reference C01070013, Tagmentase (Tn5 transposase) - loaded - replaced by the reference C01070013, Tagmentase (Tn5 transposase) - loaded - replaced by the reference C01070013, Tagmentase (Tn5 transposase) unloaded, Tagmentase (Tn5 transposase) unloaded, Tagmentase (Tn5 transposase) unloaded, Tagmentase (Tn5 transposase) unloaded, Tagmentase (Tn5 transposase) – loaded, Tagmentase (Tn5 transposase) – loaded, Tagmentase (Tn5 transposase) – loaded, Tagmentase (Tn5 transposase) – loaded, Tagmentase (Tn5 transposase) – loaded, Tagmentase (Tn5 transposase) – loaded, Tagmentase (Tn5 transposase) unloaded.
Circulomics is excited to collaborate with Diagenode to develop rapid and easy methods to facilitate the handling and shearing of HMW DNA. Combining Nanobind HMW DNA with Megaruptor shearing and Short Read Eliminator size selection allows accurate control of DNA size for long read sequencing.
Dr. Kelvin Liu, Founder and CEO of Circulomics Inc
about Megaruptor 3 DNAFluid+ Kit.
The Megaruptor 3 enables us to shear to HMW DNA to consistent and narrow size ranges. This is critical for construction of PacBio libraries and most importantly for samples with limiting amounts of DNA. The fact that it is easy to use is a significant plus in a busy lab.
Alvaro Hernandez, Ph.D., Director of the High-Throughput Sequencing and Genotyping Unit of the Roy J. Carver Biotechnology Center at the University of Illinois at Urbana-Champaign.
about Megaruptor® 3.
We were very happy with the method. It gave good results in the end, and required much smaller samples than we need to reliably perform conventional ChIP-seq.
In our view, the main advantages of the ChIPmentation kit compared to our conventional ChIP-seq protocol are (most important first):
- smaller sample requirement,
- simpler workflow with less that can go wrong,
- slightly higher resolution and signal: noise ratio.
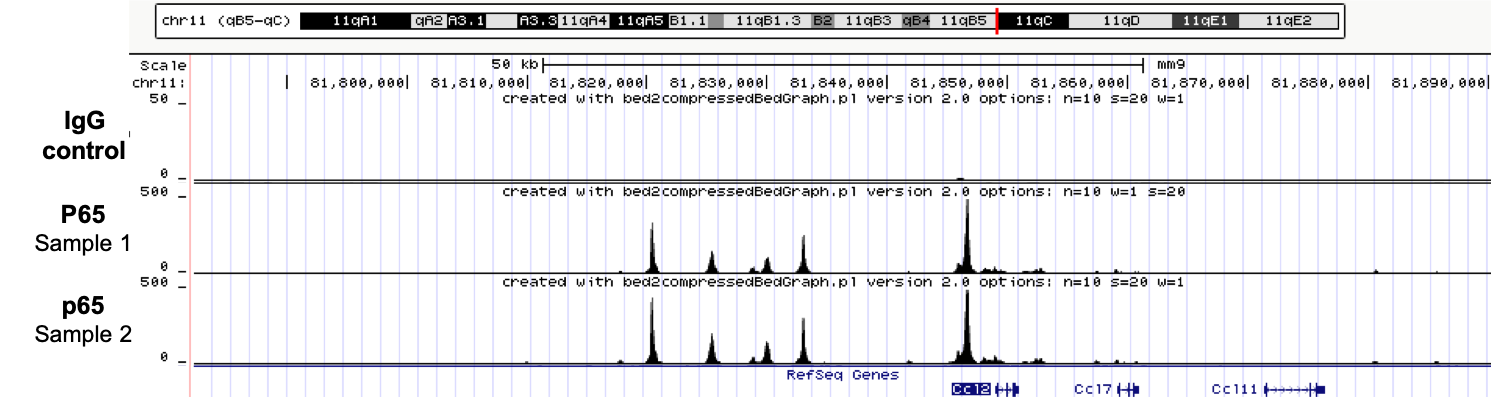
ChIPmentation sequencing profiles for p65. Chromatin preparation and immunoprecipitation have been performed on stimulated NIH3T3 cells using the iDeal ChIP-seq kit for TFs (Cat. No. C01010055). Chromatin from 4,000,000 cells was used for the immunoprecipitation in combination with either anti-p65 antibody or IgG. The library preparation was performed with the TAG Kit for ChIPmentation (Cat. No. C01011030) and 24 SI for ChIPmentation (Cat. No. C01011031).
about iDeal ChIP-seq kit for Transcription Factors, iDeal ChIP-seq kit for Transcription Factors, iDeal ChIP-seq Kit for Transcription Factors, TAG Kit for ChIPmentation.
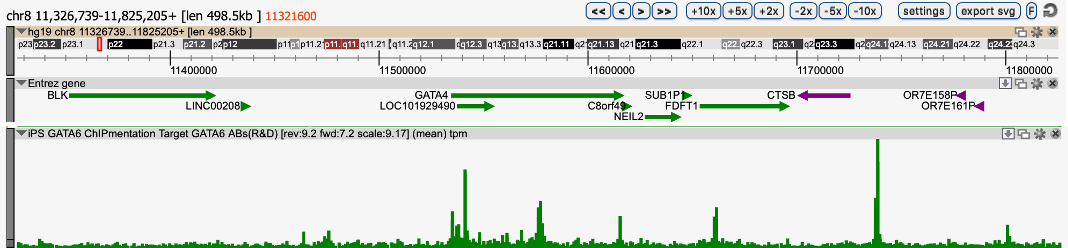
One of our issues was that we could obtain only a limited number of cells, which is not enough for canonical ChIP-seq protocols. To solve this issue, we used the Diagenode ChIPmentation solution composed of iDeal ChIP-seq Kit for Transcription Factor, TAG Kit for ChIPmentation, and 24 SI for ChIPmentation. We performed ChIPmentation with IP-Star automated system for GATA6 in 2 million GATA6-overxpressing iPS cells. The result showed clear signal/noise ratio and was highly reproducible. This solution also worked in vitro differentiated definitive endoderm cells (data not shown).
Region 1
Region 2
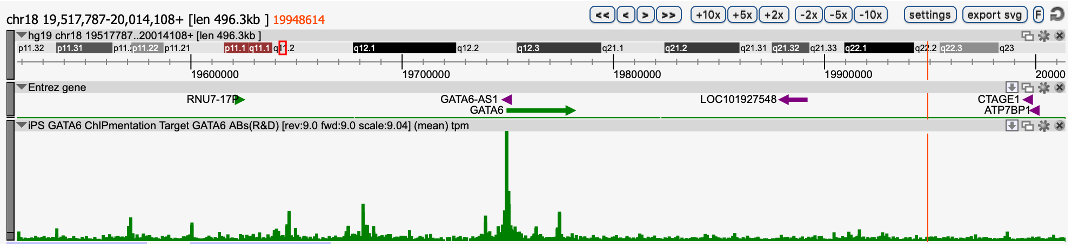
Figure 1. ChIPmentation sequencing profiles for Gata6
Chromatin preparation and immunoprecipitation have been performed on hiPSCs (human induced Pluripotent Stem Cells) overexpressing Gata6 using the iDeal ChIP-seq kit for TFs (Cat. No. C01010055). Chromatin from 2,000,000 cells was used for the immunoprecipitation in combination with either anti-GATA6 antibody. The library preparation was performed with the TAG Kit for ChIPmentation (Cat. No. C01011030) and 24 SI for ChIPmentation (Cat. No. C01011031).
about iDeal ChIP-seq kit for Transcription Factors, iDeal ChIP-seq kit for Transcription Factors, iDeal ChIP-seq Kit for Transcription Factors, TAG Kit for ChIPmentation, 24 SI for Tagmented libraries.
We at PreOmics are using the Bioruptor® Pico on a daily basis along with the iST kits for our protein sample processing workflows. The ultrasonication-based lysis greatly improves cell and tissue disruption, as well as shearing of DNA, generating samples with significantly increased peptide and protein identifications. The Bioruptor Pico in our lab has successfully processed several thousands of samples such as cells, mammalian and plant tissues, bacteria or yeast. We are highly satisfied with the handling and reproducibility of the instrument and recommend it to improve sample preparation for protein analysis.
Dr. Fabian Hosp, Head of Applications, PreOmics, Martinsried, Germany
about iST cartridge holder for the Picoruptor (PreOmics).
As a PacBio Certified Service Provider it is critical that sample processing in my laboratory is precise and reproducible. For genome sequencing projects, the fragmentation of genomic DNA to precise and reproducible sizes is essential in order to optimize conditions for library preparation, sequencing, and downstream assembly. For this my laboratory relies on the Megaruptor system. The Megaruptor is the optimal system for long DNA fragment generation and tight fragment length distribution.
Brewster Kingham, Delaware Biotechnology Institute, University of Delaware
about Megaruptor® 3.
The NGS Competence Center Tübingen (NCCT), together with four other national centers, has been established by the DFG (German Research Foundation) to support research projects with diverse needs of high-throughput sequencing technologies.
Long-read sequencing is very helpful to answer scientific questions in various topics such as microbiology or clinical research. We have noticed that the data yield of Nanopore sequencing can be notably increased by shearing the high molecular weight genomic DNA with an average size distribution of ~30kb and obtaining a read length N50 of 30kb. In this context, the Megaruptor 3 was critical to achieve long, homogenous and reproducible DNA preparation.
Megaruptor 3 is able to shear different molecular weight ranges up to 100kb; provided the input genomic DNA is of high-molecular weight. We have tested the Megaruptor 3 with genomic DNA from human blood, fibroblasts and difficult samples such as bacterial genomic DNA with high viscosity. With the Megaruptor 3 we have easily sheared up to 8 samples in parallel, saving preparation time. We have tested concentrations as low as 5 ng/µL and up to 70 ng/µL, saving sample material for optimization and meeting downstream requirements for library preparation.
Finally, handling of the Megaruptor 3 is quick, with a simple interface. Diagenode is fast in delivering consumables and these are ready-to-go. Sample preparation requires one pipetting step. You need to enter 2 parameters of your sample: volume and concentration in addition to the speed related to your desired size. It is a safe process without sample cross-contamination. It is easy to control whether the sample is going through the hydropore. The system is fast; for the concentration and speed conditions we have tested, runs were completed between half an hour and 2 hours.
Elena Buena Atienza and Dr. Nicolas Casadei, Institute of Medical Genetics and Applied Genomics, University Clinics Tübingen
about Megaruptor® 3.
I have used Diagenode's antibodies for my ChIP–qPCR experiments in HK-2 cells. The antibodies performed very well in our experiments with specific signal, and good signal to noise ratio in the promoter of the gapdh gene.
Figure 1. ChIP results obtained with the Diagenode antibody directed against H3K4me3 ChIP assays were performed using human HK-2 cells, the Diagenode antibody against H3K4me3 (Cat. No. C15410003) and optimized PCR primer pairs for qPCR. ChIP was performed with standard fixation and sonication protocol, using sheared chromatin from 4 million cells. A total amount of 4 ug of antibody were used per ChIP experiment, normal mouse IgG (4 μg/IP) was used as a negative IP control. Quantitative PCR was performed with primers specific for the promoter of the active genes GAPDH and. Figure 1 shows the recovery, expressed as a % of input (the relative amount of immunoprecipitated DNA compared to input DNA after qPCR analysis). These results are in accordance with the observation that trimethylation of K4 at histone H3 is associated with the promoters of active genes.
Claudio Cappelli PhD. Post-Doc. Investigator, Laboratory of Molecular Pathology, Institute of Biochemestry and Microbiology, University Austral of Chile, about H3K4me3 polyclonal antibody Premium, 50 μl size.
about H3K4me3 Antibody, H3K4me3 polyclonal antibody (sample size).
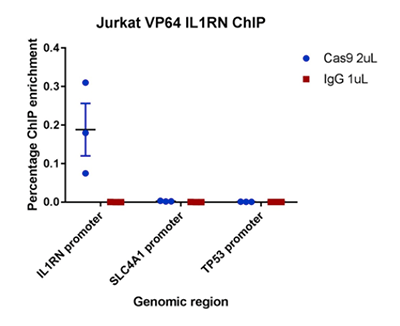
I used ChIP-qPCR with the Diagenode CRISPR/Cas9 polyclonal antibody to successfully show that Cas9 binds to the target region of my sgRNA, validating my CRISPR experiment. The antibody produced minimal background signal at non-specific genomic regions. I am now using the antibody to validate further sgRNA in different CRISPR cell lines.
ChIP was performed on Jurkat cells expressing dCas9-VP64-mCherry and a sgRNA targeting the IL1RN promoter. Each IP was performed using 4 million cells and 2 µL CRISPR/Cas9 polyclonal antibody (Diagenode C15310258) or 1 µg rabbit IgG control antibody (Diagenode C15410206). qPCR was carried out on undiluted ChIP DNA using SYBR green and PCR primers directed against the sgRNA binding site at IL1RN, as well as two non-target regions at the SLC4A1 and TP53 promoters. ChIP enrichment was measured using the percent input method.
Researcher from the University of Manchester
about CRISPR/Cas9 Antibody, CRISPR/Cas9 Antibody (sample size).
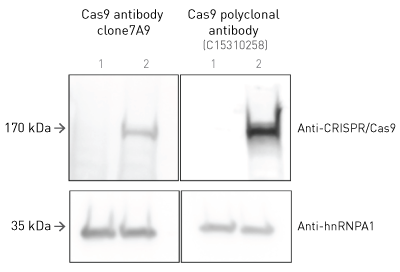
Diagenode’s CRISPR/Cas9 polyclonal antibody shows superior signal than the original clone 7A9: a researcher from EPFL in Lausanne, Switzerland has compared these two antibodies in Western blot.
Western blot was performed using HCT116 DKO cells transduced with Krab-dCas9 (2) or non-transduced (1) cells. Then, 100,000 cells were lysed in sample buffer 2x and boiled 5 min at 95°C before loading in a 15% acrylamide gel. The same sample was loaded 3x in the same gel. The membrane was cut in 3 parts for each antibody. Membrane was blocked 1h with 3% BSA at RT. Antibodies were diluted 1:1,000 in 3% BSA and incubated overnight at 4°C. Secondary incubation was done for 1h at RT (1:10,000 dilution). Anti-hnRNPA1 was used as a loading control.
EPFL in Lausanne
about CRISPR/Cas9 Antibody, CRISPR/Cas9 Antibody (sample size).
As Genomic services facility, Genoma Lab team, have a large experience, processing different samples for NGS library prep and usually those protocols start with a fragmentation step and frequently the enzimatic option works fine at the first attemp, for the majority kind of samples, Some complex samples (environmental and clinical samples) are more resistant and require more treatments with different setups, which is a problem in low amount DNA and unique samples, causing at the same time a negative impact in experimental planning, times of processing samples and cost. Because of this, the introduction of mechanical fragmentation strategy, using Diagenoder One, became more relevant in the Lab routine. Bioruptor One improved our NGS workflows, providing better quality, cost and time of experiments, with a quick, easy, versatile and clean method!
Ph.D. Carolina Sánchez & Genoma Lab team. Genomic services facility Universidad Mayor. Santiago. Chile.
about Diagenode One sonication device, 50 ?l Microfluidic Chips for the One.
We have had an excellent experience with the RRBS service provided by Diagenode. Our project was based on DNA extracted from human fresh and paraffin-embedded skeletal muscle, and the RRBS and Bioinformatic results were reliable and consistent with what we expécted. We highly recommend their service
Mario Roque - Instituto de Histología y Embriología de Mendoza (IHEM), Mendoza, Argentina
about RRBS Service (Reduced Representation Bisulfite Sequencing).
We are very happy with the services provided by Diagenode. In our project, Reduced Representation Bisulfite Sequencing (RRBS) was used and it worked well for both high-quality DNA and DNA extracted from formalin-fixed, paraffin-embedded (FFPE) material. Bioinformatic analyses were comprehensive and high-quality results were obtained. Diagenode’s services can be recommended.
Satu Mäki-Nevala, PhD, University of Helsinki
about RRBS Service (Reduced Representation Bisulfite Sequencing).
We sheared the DNA on the Diagenode One and used the MicroPlex Library Preparation v2 Kit to create DNA libraries for whole genome sequencing of four plant species for which there is no reference genome available. Previous attempts with a commercial Tn5-transposase based method gave unsatisfactory results. However, the Diagenode MicroPlex kit was quicker, easier, and gave the expected profile of fragment sizes. In just 30 seconds of sonication, we obtained a fragment distribution centered at 270 bp. The library construction took only 2 hours with this kit. The library was sequenced in a NexSeq 550 in High-Output mode, giving 85% based with>Q30.
PhD. Ricardo Verdugo, Assistant Professor, University of Chile
about Diagenode One sonication device, MicroPlex Library Prep. Kit v2 (12 indexes) - DISCONTINUED.
I like the Diagenode CATS RNA-seq kit for a number of reasons.
Jordan Lewandowski, PhD; HSCRB // Harvard
The kit is approachable for any research user. The things I care about are time, reproducibility, and ease. In my hands, the CATS mRNA-seq kit has generated highly reproducible libraries from very low input RNA that goes into polyA selection. This has significantly decreased the amount of time I spend doing library preps, enabling me to further engage with my research.
Given the low input range of up to 1 ng of our cell free plasma derived small RNA samples, commercially available kits and various published protocols failed in generating a library suitable for NGS with attempts resulting in high concentrations of adaptor dimer or failed library amplifications.
In light of this, we are more than happy to have tested the CATS small RNA-seq kit by Diagenode. Even with limited input material we got a rich library with absolutely no detectable adaptor dimer, as expected given it is a ligation free method. The kit performed superbly, in contrast to other commercially available kits or published protocols, with less hassle and with a greater ease of use.
Highly recommended for anyone who is struggling with generating a library from low input and challenging samples.
Jonatan Darr, PhD, Environmental Epigenetics Group, Institute of Experimental Genetics, Helmholtz Zentrum München, Germany
I have used Diagenode's antibodies for my ChIP Seq experiments. The antibodies performed very well in our experiments with specific signal, and good signal to noise ratio.
Antibodies used in our lab: Pol II Monoclonal Classic (C15200004).
Junaid Akhtar, Institute of Development Biology and Neurobiology, Mainz, Germany
about Pol II Antibody, Pol II Antibody (sample size).