CRISPR systems are adaptable immune mechanisms which are present in many bacteria to protect themselves from foreign nucleic acids, such as viruses, transposable elements or plasmids. The CRISPR/Cas9 (CRISPR-associated protein 9 nuclease) system from S. pyogenes was the first to be adapted for inducing sequence-specific double stranded breaks and targeted genome editing. This system is unique and flexible due to its dependence on RNA as the moiety that targets the nuclease to a desired DNA sequence and can be used to induce indel mutations, specific sequence replacements or insertions and large deletions or genomic rearrangements at any desired location in the genome. In addition, Cas9 can also be used to mediate upregulation of specific endogenous genes or to alter histone modifications or DNA methylation, Recently, the CRISPR/Cas9 from S. aureus (UniProtKB/Swiss-Prot entry J7RUA5) was also shown to be suitable for humane genome editing. The S. aureus CRISPR/Cas9 has the advantage that it’s smaller and therefore easier to transfect cells with, whereas the efficiency and specificity are similar.
S. aureus CRISPR/Cas9 Antibody
Catalog Number
Format
Price
Other format
| Lot | 10µg/50µg: 001 // 100µg: 002 |
|---|---|
| Concentration | 2 μg/μl |
| Species reactivity | Mouse |
| Type | Monoclonal |
| Purity | Protein G purified |
| Host | Mouse |
| Precautions | This product is for research use only. Not for use in diagnostic or therapeutic procedures. |
| Applications | Suggested dilution | References |
|---|---|---|
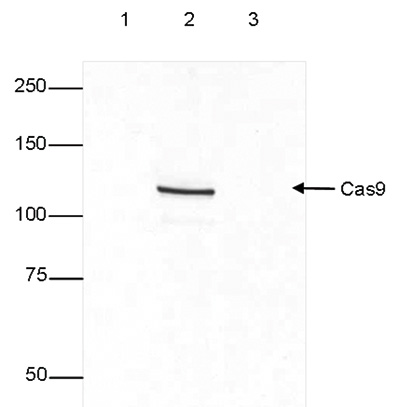
| Western Blotting | 1:4,000 | Fig 1 |
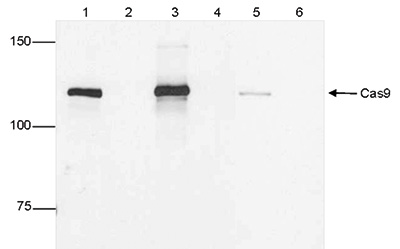
| Immunoprecipitation | 5 μg/IP | Fig 2 |
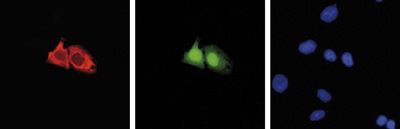
| Immunofluorescence | 1:400 | Fig 3 |