Diagenodeとは
Diagenode, a Hologic company, is a leading global provider of complete solutions for epigenetics research, biological sample preparation, and diagnostics assays. It was acquired by Hologic, Inc in March 2021.
The company has developed a comprehensive approach to gain new insights into epigenetics studies. The company offers innovative Bioruptor® shearing and IP-Star® automation instruments, reagent kits, and high quality antibodies to streamline DNA methylation, ChIP, and ChIP-seq workflows. The company's latest innovations include a unique, full automation system, the industry's most validated antibodies, the Megaruptor shearing system for long fragment generation in sequencing, and epigenetics assay services.
At Diagenode, our goal is to build products with pride and the highest level of performance. Our team of epigenetics experts develop products by getting feedback from our customers as well as the scientific and medical communities around us. We strive to develop superior and easy-to-use products to bring epigenetics research and diagnostics to new frontiers.
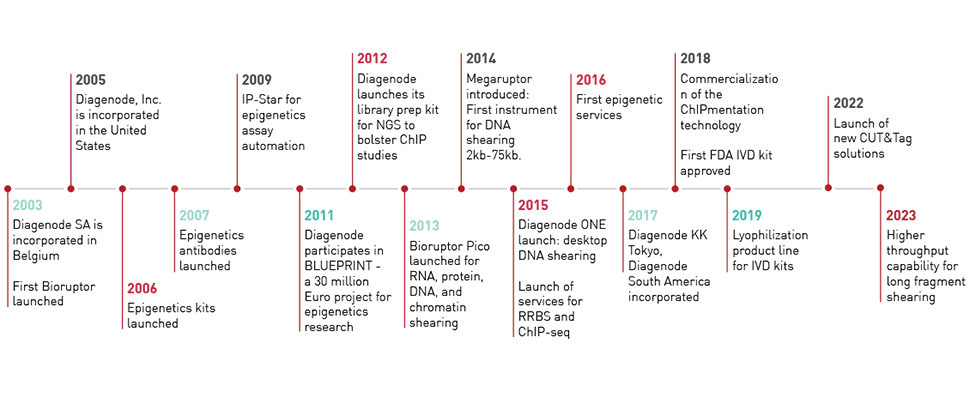
エピジェネティクス研究
Epigeneticsは、生命科学研究において革命的な役割を果たしています。 Diagenodeは絶えず研究における問題への解決案を探求し、市場でも最高品質のエピジェネティクスツールを提供いたします。
当社の詳細については、お問い合わせください:
- Europe: custsupport@diagenode.com
- North America: custsupport.na@diagenode.com