关于我们
Diagenode, a Hologic company, is a leading global provider of complete solutions for epigenetics research, biological sample preparation, and diagnostics assays. It was acquired by Hologic, Inc in March 2021.
The company has developed a comprehensive approach to gain new insights into epigenetics studies. The company offers innovative Bioruptor® shearing and IP-Star® automation instruments, reagent kits, and high quality antibodies to streamline DNA methylation, ChIP, and ChIP-seq workflows. The company's latest innovations include a unique, full automation system, the industry's most validated antibodies, the Megaruptor shearing system for long fragment generation in sequencing, and epigenetics assay services.
At Diagenode, our goal is to build products with pride and the highest level of performance. Our team of epigenetics experts develop products by getting feedback from our customers as well as the scientific and medical communities around us. We strive to develop superior and easy-to-use products to bring epigenetics research and diagnostics to new frontiers.
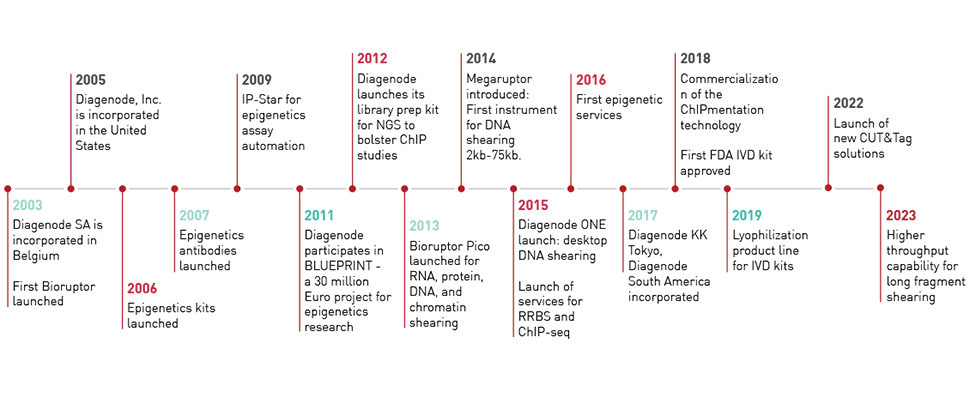
表观遗传学研究
表观遗传学在生命科学研究中发挥着革命性的作用。Diagenode 不断创新解决方案,并提供最高质量的表观遗传学工具。
有关我们公司的更多信息,请联系我们:
- Europe: custsupport@diagenode.com
- North America: custsupport.na@diagenode.com